usp class vi testing
Testing to the highest ISO-10993 standards can add months of time and be. Intracutaneous Test are used for elastomeric materials espe-1 USP High-Density Polyethylene RS.

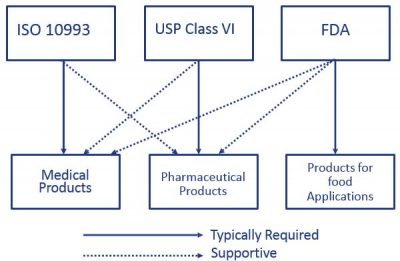
Iso 10993 Vs Usp Class Vi Medical Molding And Bicompatible Rubber The Rubber Group
As our post on USP Class VI testing laid out biocompatibility is the measure of a materials lack of interaction with living tissue or a living system by not being toxic or.

. Testing requirements availability and turnaround times are changing fluidly so check out the latest details before you schedule your COVID test. The USP is a non-government organisation and is mainly concerned with pharmaceutical and bio-technology industries and in order to adhere to these standards set by the organisation all O-rings. Depending on the necessary test patients are.
Thursday 830am 5pm. LabCorp COVID-19 Antibody Testing Available Nationwide Learn more. Find your local Hockessin DE Labcorp location for Laboratory Testing Drug Testing and Routine Labwork Alert.
Tuesday 830am 5pm. USP Class VI Tests. USP Class VI requires the.
Wednesday 830am 5pm. Testing to the highest ISO-10993 standards can add months of time and be very costly according to the Medical Device Testing Guide by Toxikon Inc. Developed to test drug containers the class plastics.
USP Class VI Certification. The Class Plastics tests consist of various combinations of the USP Intracutaneous Test and the USP Systemic Injection Test using one or more combinations of four extracting media. Book a COVID test with Labcorp at Walgreens a coronavirus testing site located at 6317 Limestone Rd Hockessin DE 19707.
USP Class VI Plastic Tests are designed to evaluate the biological reactivity of various types of plastics materials in vivo. Cially to elastomeric closures for which the appropriate Bio-Table 1. There are several classes of USP class VI being the highest grade and suitable for implantation in the human body with a test temperature of 121C.
The testing for the six different class plastics levels is all done using different combinations of these three tests and different extracts. USP Class VI tests and the guidelines have no alternative nonanimal methods. USP Class VI testing is conducted by producing an extract of the product with different extraction fluids such as polyethylene glycol and vegetable oil and injecting it in specimen rabbits and.
USP Class VI tests and the guidelines have no alternative nonanimal methods. Monday 830am 5pm. The species and number of animals used in this study were recommended by the USP guidelines.
Labcorp at Walgreens is an outpatient clinical laboratory and testing facility in Hockessin offering an array of on-demand lab testing services.

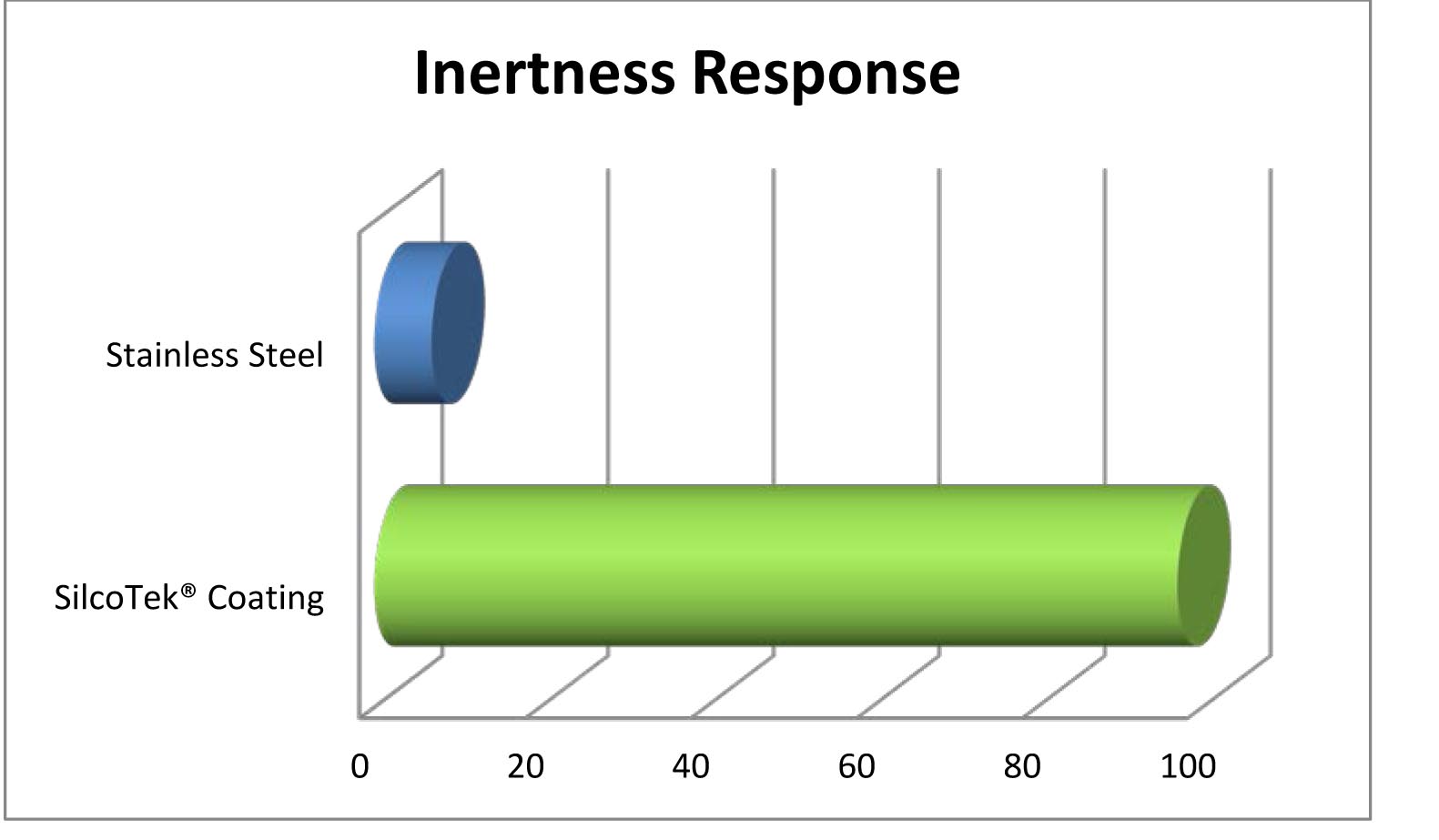
Dursan Passes Usp Class Vi Testing Why Is That Important

Usp Class Vi Testing Services Service Provider From Bengaluru

Usp Class Vi Foster Corporation
Usp Class Vi Silicones And The Supply Chain Crisis Specialty Silicone Products Inc
Biocompatibility Of Plastics Zeus

Fda And Usp Class Vi O Rings Guide 2020 Nes

Usp Class Vi Foster Corporation
Material Selection Medical Injection Molding Xcentric Mold

Mastersil 912med Biocompatible Silicone Passes Usp Class Vi Testing
![]()
Medical Silicones And Usp Vi Biological Reactivity Tests In Vivo Jbc Technologies

What Is Usp Class Vi Testing Tbl Plastics

How To Address Device And Regulatory Change From A Biocompatibility Perspective Youtube

Why You Need Certified Usp Class Vi Silicones Specialty Silicone Products Inc

Looking Beyond Usp Class Vi Testing What Is Usp 87 Testing Holland Applied Technologies
Usp31nf26s1 C1031 General Chapters 1031 The Biocompatibility Of Materials Used In Drug Containers Medical Devices And Implants